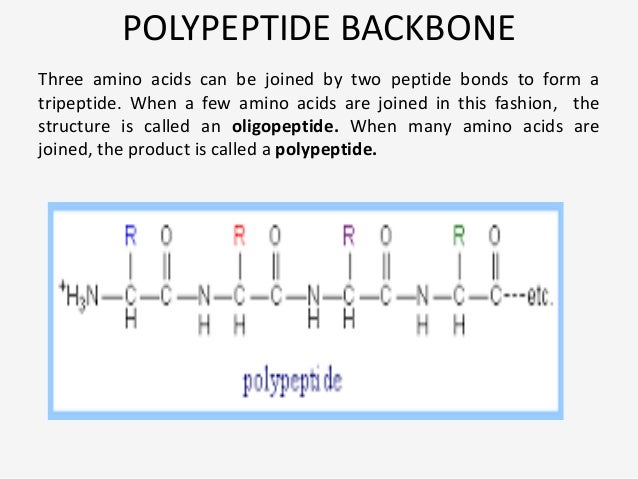
Output example: aag000_0001.m.pdb~ and with the TER added, aag000_0001.m.pdb Input: gpep_nat.pdb, list_of_pdbs (example of pdbs in list - aag000_0001.pdb) (NOTE: When building with centroid mode, we don't use a TER inīetween chains A and B. Have to repack all of the gpep1.pdb positions during the fullatom Position 342 and put this # after "tail -". For example, if you start the design at sequence positionģ42, like we do here, check your gpep_nat.pdb file (original all-atom pdbįile) for the line # for the last atom in sequence position 341 and put this in after "head -", then check your centroid files for the first atom at You will need to edit merge_pdb.csh if you want to change which residues areīeing merged.
Gpep1.pdb) using merge_pdb.csh like this: merge_pdb.csh gpep1_nat.pdb. Merge centroid designs with fullatom starting structure (in this case Input: g000.loops, g000.cst (I didn't use constraints) Run command line: rosetta.mactel aa input_pdb _ -s g000.pdb -loops An example of a constraint file isĬopy over starting structure g000.pdb from step 2. In other words, part 2)Ībove can generate a pdb file with all alanines in the region that willīe designed OR the sequence can be specified with a fasta file (seeĪbove) so that big hydrophobics fall in buried regions and hydrophilicsįall in solvent exposed regions, etc. Location OR direct the redesigned peptide toward the desired orientation Make constraint file to make the designed region fall into the desired Number being the sequence position where the design will end. In the loop file, the 1st numer is the # of positions to beīuilt, second number is sequence position to start design, with the final Make a loop file to specity what residues can move. Input: g000.zones, g000_.fasta, gpep_nat.pdb With no "TER" between chains A and B, and the numbering is sequential But my modified pdb (gpep_nat.pdb) starts with position 1, "Rosetta numbers", so the original pdb starts with sequence position 30,Ĭontains a "TER" between chain A and B, and chain B starts with sequence NOTE: The input file has the sequence positions re-numbered in Removed from all sequence positions, and the region that will be redesigned The resulting file should look something like g000.pdb. For example: -zonesfile g000.zones -fastafile g000_.fasta -parentpdb gpep_nat.pdb -outpdb g000.pdb Sequence improved the quality of the centroid models more effectively than IĪlso included a fasta file (see g000_.fasta) because I felt that setting the Make starting structure using and the g000.zones file. This protocol uses 2 separate rosetta runs - one is centroid mode to buildīackbone coordinates and the other is a design run to find a low-energy Rosetta.mactel -design -l list_of_pdbs -tail -begin 342 -end 351 -chain_ -series bb -protein g000 -resfile g000_resfile -ex1 -ex2 -extrachi_cutoff 1 -exOH -no_his_his_pairE -tight_hb -try_both_his_tautomers Running the protocol Important flags: -ex1, -ex2, -exOH, -extrachi_cutoff 1 all seem to be very important for the sequence design run.Įxample Rosetta Command Line: rosetta.mactel aa input_pdb _ -s g000.pdb -loops Protein, then designs a low-energy sequence. This protocol builds (or extends) a backbone for a peptide bound to a target GoLoco was confirmed using disruptive mutations at the Gi1:GoLoco interface,Īnd the correctness of the computational design was assessed with by x-ray

The redesigned GoLoco binds to Gi1 with aĭissociation constant of 810nM, the correct binding of the newly designed We removed this C-terminal portion of GoLoco, replacing the random coil With the C-terminal random coil region binding to the all-helical domain of Our model system is an x-ray crystal structure of Gi1 bound to the Our project is the computational design of a new high-affinity protein-protein Mischa Machius, David Siderovski, Brian Kuhlman, Kuhlman lab, 2010, Session 3 Redesigned RGS14 GoLoco, Deanne Sammond, Dustin Bosch, Glenn Butterfoss,
